2026 / 2025 / 2024 / 2023 / 2022 / 2021 / 2020 / 2019 / 2018 / 2017 / 2016 / 2015 / 2014 / 2013 / 2012 / others (PDF)
2026
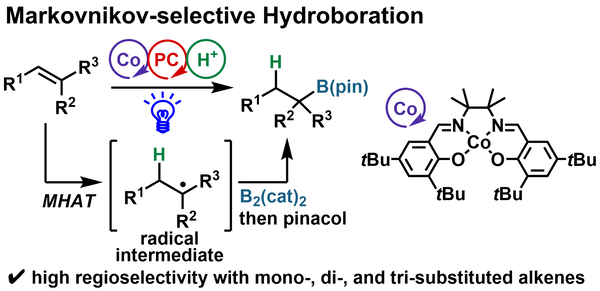
Markovnikov-Selective Hydroboration of Unactivated Alkenes via MHAT under Cobalt/Photoredox Dual Catalysis
Seino, Y.; Dozen, S.; Yoshino, T.; Higashida, K.; Matsunaga, S.
Org. Lett. 2026, ASAP. DOI: 10.1021/acs.orglett.6c00348
[KURENAI]
Synthesis of Sulfonyl Fluorides from Tertiary Sulfonamides via N-Methylation Using Me-λ3-Iodane
Watanabe, T.; Katsuta, R.; Higashida, K.; Yoshino, T.; Matsunaga, S.
Synlett 2026, e-first. DOI: 10.1055/a-2782-6367
[KURENAI]
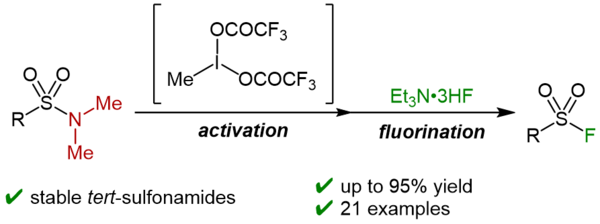
Tetranuclear Gold(I) Complexes Bearing Imidazo[1,5-b]pyridazin-7-ylidene Ligands: Synthesis and Catalytic Applications in Nucleophilic Addition to Alkynes
Yamamoto, I.; Yoshino, T.; Higashida, K.; Matsunaga, S.
Organometallics 2026, 45, 204-211. DOI: 10.1021/acs.organomet.5c00453
[KURENAI]
2025
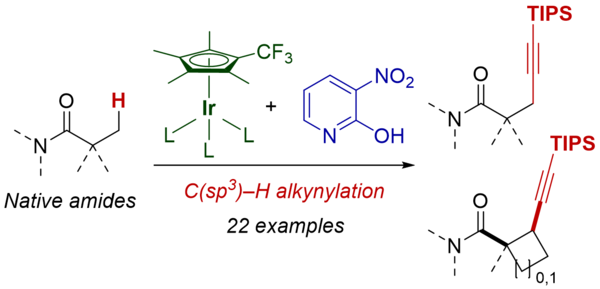
Native Amide-Directed C(sp3)–H Alkynylation Using an Electron-Deficient Iridium Catalyst with Pyridone Ligand Assistance
Kimura, S.; Kobayashi, T.; Hirata, Y.; Higashida, K.; Yoshino, T.; Matsunaga, S.
Org. Lett. 2025, 27, 13276-13280. DOI: 10.1021/acs.orglett.5c04262
[KURENAI]
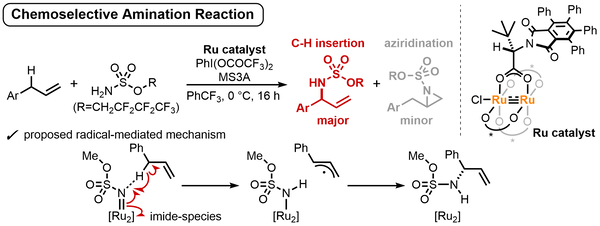
Enantioselective C(sp3)–H Amination of Allyl Benzene Derivatives Enabled by Paddle-wheel Diruthenium Catalysis
Mori, K.; Makino, K.; Kosaka, M.; Yoshino, T.; Higashida, K.; Kurashige, Y.; Matsunaga, S.
Bull. Chem. Soc. Jpn. 2025, 98, uoaf106. DOI: 10.1093/bulcsj/uoaf106
[KURENAI]
1,2-Dihydro-1,2,4,5-tetrazine-3,6-dione (TETRAD)
Yoshino, T.; Matsunaga, S.
Encyclopedia of Reagents for Organic Synthesis (e-EROS) DOI: 10.1002/047084289X.rn02433
In Situ Generation of Alkyl–λ3–Iodanes as Soft and Highly Reactive Electrophiles
Katsuta, R.; Watanabe, T.; Matsuoka, K.; Watanabe, K.; Higashida, K.; Yoshino, T.; Matsunaga, S.
ChemistryEurope 2025, 3, e202500154. DOI: 10.1002/ceur.202500154
[KURENAI]
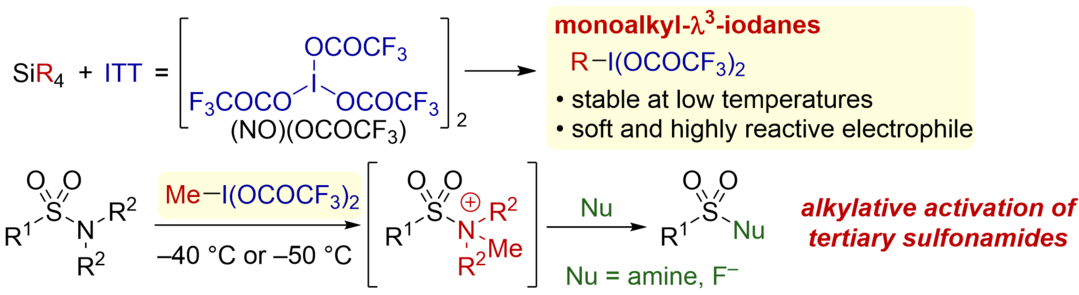
Distal Heteroaryl Ipso-Migration of Unactivated Alkenes via Cobalt/Photoredox-Catalyzed Hydrogen/Deuterium Atom Transfer
Yamashita, M.; Suzuki, A.; Yoshino, T.; Higashida, K.; Kojima, M.; Matsunaga, S.
Chem. Asian. J. 2025, 20, e00448. DOI: 10.1002/asia.202500448
[KURENAI]
Enantioselective C–H Amidation of Sulfondiimines for the Synthesis of 1,2,4-Benzothiadiazine-1-Imines under Cobalt Catalysis
Murata, A.; Endo, T.; Hirata, Y.; Higashida, K.; Yoshino, T.; Matsunaga, S.
Org. Chem. Front. 2025, 12, 4236-4241. DOI: 10.1039/D5QO00355E
[KURENAI]
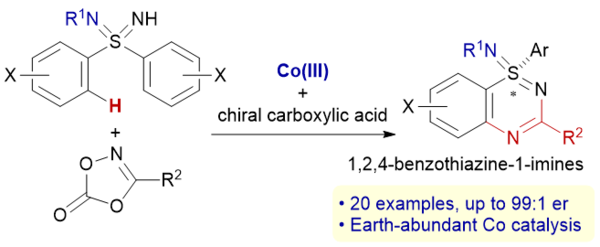
Multiple Deuterium Atom Transfer Perdeuteration of Unactivated Alkenes under Base-Assisted Cobalt/Photoredox Dual Catalysis
Suzuki, A.; Higashida, K.; Yoshino, T.; Matsunaga, S.
Angew. Chem., Int. Ed. 2025, 64, e202500233. DOI: 10.1002/anie.202500233
[KURENAI]
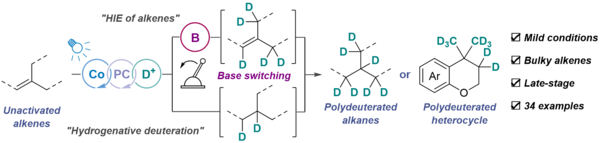
Enantioselective Intermolecular Benzylic C–H Amination under Chiral Paddle-Wheel Diruthenium Catalysis
Makino, K.; Mori, K.; Kiryu, S.; Miyazawa, T.; Kumagai, Y.; Higashida, K.; Kojima, M.; Yoshino, T.; Matsunaga, S.
ACS Catal. 2025, 15, 523-528. DOI: 10.1021/acscatal.4c06504
[KURENAI]
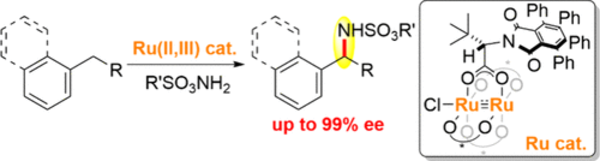
Site-selective C(sp3)–H and Switchable C(sp3)–H/C(sp2)–H Functionalization Enabled by Electron-deficient Cp*CF3Ir(III) Catalyst and Photosensitizer
Hirata, Y.; Kimura, S.; Higashida, K.; Yoshino, T.; Matsunaga, S.
Angew. Chem., Int. Ed. 2025, 64, e202421026. DOI: 10.1002/anie.202421026
[KURENAI]
Highlighted in Synfacts.
2024
Cobalt/Photoredox Dual-Catalyzed Cross-Radical Coupling of Alkenes via Hydrogen Atom Transfer and Homolytic Substitution
Yamaguchi, Y.; Hirata, Y.; Higashida, K.; Yoshino, T.; Matsunaga, S.
Org. Lett. 2024, 26, 4893-4897. DOI: 10.1021/acs.orglett.4c01370
[KURENAI]

2023
Enantioselective Synthesis of 1,2-Benzothiazine 1-Imines via RuII/Chiral Carboxylic Acid-Catalyzed C–H Alkylation/Cyclization
Huang, L.-T; Kitakawa, Y.; Yamada, K.; Kamiyama, F.; Kojima, M.; Yoshino, T.; Matsunaga, S.
Angew. Chem., Int. Ed. 2023, 62, e202305480. DOI: 10.1002/anie.202305480

Synthesis of Benzo[c]azepine-1,3(2H)-diones via C–H Alkylation/Cyclization with α,β-Unsaturated Acyl Fluorides
Hosoi, S.; Hirata, Y.; Kurihara, T.; Kojima, M.; Yoshino, T.; Matsunaga, S.
Asian J. Org. Chem. 2023, 12, e202300218. DOI: 10.1002/ajoc.202300218

Catalytic Enantioselective Amination of Enol Silyl Ethers Using a Chiral Paddle-Wheel Diruthenium Complex
Makino, K.; Kumagai, Y.; Yoshino, T.; Kojima, M.; Matsunaga, S.
Org. Lett. 2023, 25, 3234-3238. DOI: 10.1021/acs.orglett.3c00940

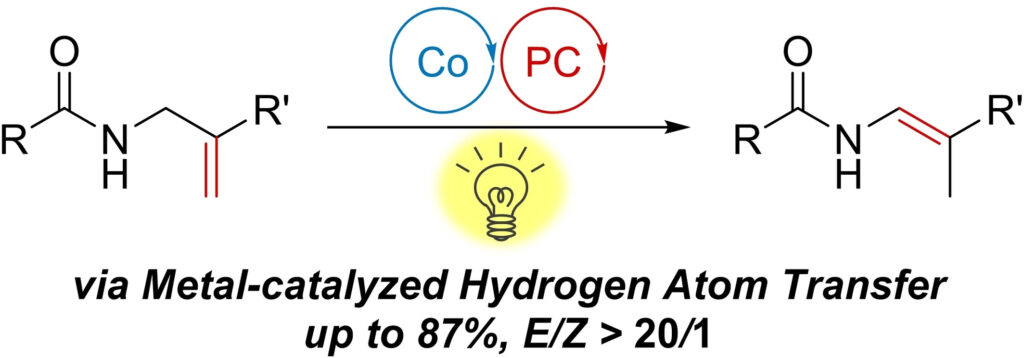
Synthesis of Polysubstituted Enamides by Hydrogen Atom Transfer Alkene Isomerization Using Dual Cobalt/Photoredox Catalysis
Seino, Y.; Yamaguchi Y.; Suzuki, A.; Yamashita, Y.; Kamei, Y.; Kamiyama, F.; Yoshino, T.; Kojima, M.; Matsunaga, S.
Chem. Eur. J. 2023, 29, e202300804. DOI: 10.1002/chem.202300804
Formation of Isolable Dearomatized [4 + 2] Cycloadducts from Benzenes, Naphthalenes, and N-Heterocycles Using 1,2-Dihydro-1,2,4,5-tetrazine-3,6-diones as Arenophiles under Visible Light Irradiation
Ikeda, K.; Kojima, R.; Kawai, K.; Murakami, T.; Kikuchi, T.; Kojima, M.; Yoshino, T.; Matsunaga, S.
J. Am. Chem. Soc. 2023, 145, 9326-9333. DOI: 10.1021/jacs.3c02556

An Electron-Deficient CpE Iridium(III) Catalyst: Synthesis, Characterization, and Application to Ether-Directed C–H Amidation
Tomita, E.; Kojima, M.; Nagashima, Y.; Tanaka, K.; Sugiyama, H.; Segawa, Y.; Furukawa, A.; Maenaka, K.; Maeda, S.; Yoshino, T.; Matsunaga, S.
Angew. Chem., Int. Ed. 2023, 62, e202301259. DOI: 10.1002/anie.202301259

Iron/Photosensitizer-Catalyzed Directed C–H Activation Triggered by the Formation of an Iron Metallacycle
Kato, Y.; Yoshino, T.; Matsunaga, S.
ACS Catal. 2023, 13, 4552-4559. DOI: 10.1021/acscatal.3c00381

Noble-Metal-Free C–H Allylation of Tetrahydroisoquinolines Using a Cobalt-Organophotoredox Dual Catalyst System
Sato, S.; Sasaki, W.; Sekino, T.; Yoshino, T.; Kojima, M.; Matsunaga, S.
Chem. Pharm. Bull. 2023, 71, 79-82. DOI: 10.1248/cpb.c22-00520

Photocatalytic Deuterium Atom Transfer Deuteration of Electron-Deficient Alkenes with High Functional Group Tolerance
Suzuki, A.; Kamei, Y.; Yamashita, M.; Seino, Y.; Yamaguchi, Y.; Yoshino, T.; Kojima, M.; Matsunaga, S.
Angew. Chem., Int. Ed. 2023, 62, e202214433. DOI: 10.1002/anie.202214433

Photo-Induced Nickel-Catalyzed Carbon-Heteroatom Coupling
Luo, H.; Wang, G.; Feng, Y.; Zheng, W.; Kong, L.; Ma, Y.; Matsunaga, S. Lin, L.
Chem. Eur. J. 2023, 29, e202202385. DOI: 10.1002/chem.202202385

2022
Native Amide-Directed C(sp3)–H Amidation Enabled by Electron-Deficient Rh(III) Catalyst and Electron-Deficient 2-Pyridone Ligand
Wakikawa, T.; Sekine, D.; Murata, Y.; Bunno, Y.; Kojima, M.; Nagashima, Y.; Tanaka, K.; Yoshino, T.; Matsunaga, S.
Angew. Chem., Int. Ed. 2022, 61, e202213659. DOI: 10.1002/anie.202213659

Iron/Photosensitizer Hybrid System Enables the Synthesis of Polyaryl-Substituted Azafluoranthenes
Kato, Y.; Yoshino, T.; Gao, M.; Hasegawa, J.; Kojima, M.; Matsunaga, S.
J. Am. Chem. Soc. 2022, 144, 18450-18458. DOI: 10.1021/jacs.2c06993

Light-Promoted Nickel-Catalyzed Aromatic Halogen Exchange
Feng, Y.; Luo, H.; Zheng, W.; Matsunaga, S. Lin, L.
ACS Catal. 2022, 12, 11089-11096. DOI: 10.1021/acscatal.2c03354

Design and Synthesis of Cyclopropane Congeners of Resolvin E3, an Endogenous Pro-Resolving Lipid Mediator, as Its Stable Equivalents
Arai, S.; Fujiwara, K.; Kojima, M.; Aoki-Saito, H.; Yatomi, M.; Saito, T.; Koga, Y.; Fukuda, H.; Watanabe, M.; Matsunaga, S.; Hisada, T.; Shuto, S.
J. Org. Chem. 2022, 87, 10501-10508. DOI: 10.1021/acs.joc.2c01110

Enantioselective C–H Functionalization Using High-Valent Group 9 Metal Catalysts
Yoshino, T.
Bull. Chem. Soc. Jpn. 2022, 95, 1280-1288. DOI: 10.1246/bcsj.20220168

Cobalt(III)/Chiral Carboxylic Acid-Catalyzed Enantioselective Synthesis of Benzothiadiazine-1-Oxides via C–H Activation
Hirata, Y.; Sekine, D.; Kato, Y.; Lin, L.; Kojima, M.; Yoshino, T.; Matsunaga, S.
Angew. Chem., Int. Ed. 2022, 61, e202205341. DOI: 10.1002/anie.202205341

Achiral Cp*Rh(III)/Chiral Lewis Base Cooperative Catalysis for Enantioselective Cyclization via C–H Activation
Kurihara, T.; Kojima, M.; Yoshino, T.; Matsunaga, S.
J. Am. Chem. Soc. 2022, 144, 7058-7065. DOI: 10.1021/jacs.2c01223

Intramolecular Hydrogen Atom Transfer Hydroarylation of Alkenes toward δ-Lactams Using Cobalt-Photoredox Dual Catalysis
Yamaguchi, Y.; Seino, Y.; Suzuki, A.; Kamei, Y.; Yoshino, T.; Kojima, M.; Matsunaga, S.
Org. Lett. 2022, 24, 2441-2445. DOI: 10.1021/acs.orglett.2c00700

Regioselective Deaminative Allylation of Aliphatic Amines via Dual Cobalt and Organophotoredox Catalysis
Sekino, T.; Sato, S.; Yoshino, T.; Kojima, M.; Matsunaga, S.
Org. Lett. 2022, 24, 2120-2124. DOI: 10.1021/acs.orglett.2c00319

1,2-Disubstituted 1,2-Dihydro-1,2,4,5-tetrazine-3,6-dione as a Dynamic Covalent Bonding Unit at Room Temperature
Kawai, K.; Ikeda, K.; Sato, A.; Kabasawa, A.; Kojima, M.; Kokado, K.; Kakugo, A.; Sada, K.; Yoshino, T.; Matsunaga, S.
J. Am. Chem. Soc. 2022, 144, 1370-1379. DOI: 10.1021/jacs.1c11665

Cp*Rh(III)/Boron Hybrid Catalysis for Directed C–H Addition to β-Substituted α,β-Unsaturated Carboxylic Acids
Tanaka, R.; Hirata, Y.; Kojima, M.; Yoshino, T.; Matsunaga, S.
Chem. Commun. 2022, 58, 76-79. DOI: 10.1039/d1cc05956d

Ru(II)/Chiral Carboxylic Acid-Catalyzed Enantioselective C–H Functionalization of Sulfoximines
Huang, L.-T.; Hirata, Y.; Kato, Y.; Lin, L.; Kojima, M.; Yoshino, T.; Matsunaga, S.
Synthesis 2022, 54, 4703-4710. DOI: 10.1055/a-1588-0072

2021
Cp*RhIII/Chiral Disulfonate/CuOAc Catalyst System for the Enantioselective Intramolecular Oxyamination of Alkenes
Hirose, J.; Wakikawa, T.; Satake, S.; Kojima, M.; Hatano, M.; Ishihara, K.; Yoshino, T.; Matsunaga, S.
ACS Catal. 2021, 11, 15187-15193. DOI: 10.1021/acscatal.1c04699

Cp*Ir(III)/Chiral Carboxylic Acid-Catalyzed Enantioselective C–H Alkylation of Ferrocene Carboxamides with Diazomalonates
Mou, Q.; Zhao, R.; Niu, R.; Fukagawa, S.; Shigeno, T.; Yoshino, T.; Matsunaga, S.; Sun, B.
Org. Chem. Front. 2021, 8, 6923-6930. DOI: 10.1039/D1QO01344K

Transition-metal-free Nucleophilic 211At-astatination of Spirocyclic Aryliodonium Ylides
Matsuoka, K.; Obata, H.; Nagatsu, K.; Kojima, M.; Yoshino, T.; Ogawa, M.; Matsunaga, S.
Org. Biomol. Chem. 2021, 19, 5525-5528. DOI: 10.1039/D1OB00789K

Chiral Carboxylic Acid Assisted Enantioselective C–H Activation with Achiral CpxMIII (M = Co, Rh, Ir) Catalysts
Yoshino, T.; Matsunaga, S.
ACS Catal. 2021, 11, 6455-6466. DOI: 10.1021/acscatal.1c01351

Development of Pseudo-C2-symmetric Chiral Binaphthyl Monocarboxylic Acids for Enantioselective C(sp3)–H Functionalization Reactions under Rh(III) Catalysis
Kato, Y.; Lin, L.; Kojima, M.; Yoshino, T.; Matsunaga, S.
ACS Catal. 2021, 11, 4271-4277. DOI: 10.1021/acscatal.1c00765
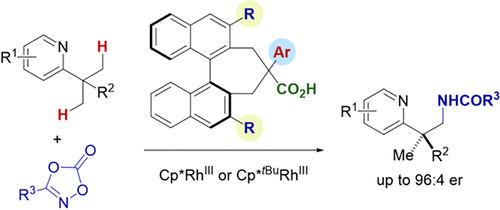
Silane- and Peroxide-Free Hydrogen Atom Transfer Hydrogenation Using Ascorbic Acid and Cobalt-Photoredox Dual Catalysis
Kamei, Y.; Seino, Y.; Yamaguchi, Y.; Yoshino, T.; Maeda, S.; Kojima, M.; Matsunaga, S.
Nat. Commun. 2021, 12, 966. DOI: 10.1038/s41467-020-20872-z

Metal-Containing Schiff Base/Sulfoxide Ligands for Pd(II)-Catalyzed Asymmetric Allylic C–H Aminations
Bunno, Y.; Tsukimawashi, Y.; Kojima, M.; Yoshino, T.; Matsunaga, S.
ACS Catal. 2021, 11, 2663-2668. DOI: 10.1021/acscatal.0c05261

Chemoselective Cleavage of Si–C(sp3) Bonds in Unactivated Tetraalkylsilanes Using Iodine Tris(trifluoroacetate)
Matsuoka, K.; Komami, N.; Kojima, M.; Mita, T.; Suzuki, K.; Maeda, S.; Yoshino, T.; Matsunaga, S.
J. Am. Chem. Soc. 2021, 143, 103-108. DOI: 10.1021/jacs.0c11645

Generation of Monoaryl-λ3-iodanes from Arylboron Compounds through ipso-Substitution
Nakano, A.; Okabe, Y.; Matsuoka, K.; Komami, N.; Watanabe, K.; Kojima, M.; Yoshino, T.; Matsunaga, S.
Heterocycles 2021, 103, 670-677. DOI: 10.3987/COM-20-S(K)45

2020
Rhodium(III)/Chiral Carboxylic Acid Catalyzed Enantioselective C(sp3)–H Alkylation of 8-Ethylquinolines with α,β-Unsaturated Carbonyl Compounds
Huang, L.-T.; Fukagawa, S.; Kojima, M.; Yoshino, T.; Matsunaga, S.
Org. Lett. 2020, 22, 8256-8260. DOI: 10.1021/acs.orglett.0c02872

Chiral Paddle-Wheel Diruthenium Complexes for Asymmetric Catalysis
Miyazawa, T.; Suzuki, T.; Kumagai, Y.; Takizawa, K.; Kikuchi, T.; Kato, S.; Onoda, A.; Hayashi, T.; Kamei, Y.; Kamiyama, F.; Anada, M.; Kojima, M.; Yoshino, T.; Matsunaga S.
Nature Catalysis 2020, 3, 851-858. DOI: 10.1038/s41929-020-00513-w

Allyl 4-Chlorophenyl Sulfone as a Versatile 1,1-Synthon for Sequential α-Alkylation/Cobalt-Catalyzed Allylic Substitution
Sekino, T.: Sato, S.; Kuwabara, K.; Takizawa, K.; Yoshino, T.; Kojima, M.; Matsunaga, S.
Synthesis 2020, 52, 1934-1946. DOI: 10.1055/s-0040-1707524
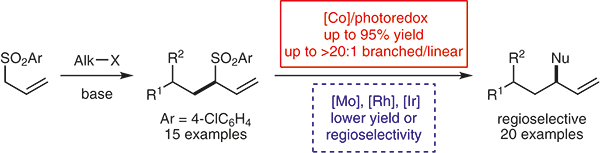
Iridium(III) Catalysts with an Amide‐Pendant Cyclopentadienyl Ligand: Double Aromatic Homologation Reactions of Benzamides by Fourfold C−H Activation
Tomita, E.; Yamada, K.; Shibata Y.; Tanaka K.; Kojima, M.; Yoshino, T.; Matsunaga, S.
Angew. Chem., Int. Ed. 2020, 59, 10474-10478. DOI: 10.1002/anie.202003009

The Merger of Photoredox and Cobalt Catalysis
Kojima, M.; Matsunaga, S.
Trends Chem. 2020, 2, 410-426. DOI: 10.1016/j.trechm.2020.01.004

Diverse Approaches for Enantioselective C–H Functionalization Reactions Using Group 9 CpxMIII Catalysts
Yoshino, T.; Satake, S.; Matsunaga, S.
Chem. Eur. J. 2020, 26, 7346-7357. DOI: 10.1002/chem.201905417

Cp*CoIII/Chiral Carboxylic Acid-Catalyzed Enantioselective 1,4-Addition Reactions of Indoles to Maleimides
Kurihara, T.; Kojima, M.; Yoshino, T.; Matsunaga, S.
Asian J. Org. Chem. 2020, 9, 368-371. DOI: 10.1002/ajoc.201900565

2019
Catalytic Enantioselective Methylene C(sp3)–H Amidation of 8-Alkylquinolines Using Cp*RhIII/Chiral Carboxylic Acid System
Fukagawa, S.; Kojima, M.; Yoshino, T.; Matsunaga, S.
Angew. Chem., Int. Ed. 2019, 58, 18154-18158. DOI: 10.1002/anie.201911268
Highlighted in Synfacts.

Imidate as the Intact Directing Group for the Cobalt-Catalyzed C–H Allylation
Tanaka, R.; Tanimoto, I.; Kojima, M.; Yoshino, T.; Matsunaga, S.
J. Org. Chem. 2019, 84, 13203-13210. DOI: 10.1021/acs.joc.9b01972

Chiral 2-Aryl Ferrocene Carboxylic Acids for the Catalytic Asymmetric C(sp3)–H Activation of Thioamides
Sekine, D.; Ikeda, K.; Fukagawa, S.; Kojima, M.; Yoshino, T.; Matsunaga, S.
Organometallics 2019, 38, 3921-3926. DOI: 10.1021/acs.organomet.9b00407

Cobalt-catalyzed Synthesis of Homoallylic Amines from Imines and Terminal Alkenes
Tanaka, R.; Kojima, M.; Yoshino, T.; Matsunaga, S.
Chem. Lett. 2019, 48 1046-1049. DOI: 10.1246/cl.190378

Cp*CoIII-Catalyzed C–H Functionalization and Asymmetric Reactions Using External Chiral Sources
Yoshino, T.; Matsunaga, S.
Synlett 2019, 30, 1384-1400. DOI: 10.1055/s-0037-1611814

C–H γ,γ,γ-Trifluoroalkylation of Quinolines via Visible-Light-Induced Sequential Radical Additions
Kumagai, Y.; Murakami, N.; Kamiyama, F.; Tanaka, R.; Yoshino, T.; Kojima, M.; Matsunaga, S.
Org. Lett. 2019, 21, 3600-3605. DOI: 10.1021/acs.orglett.9b01015

Synthesis of Heteroaryl Iodanes(III) via ipso-Substitution Reactions Using Iodine Triacetate Assisted by HFIP
Matsuoka, K.; Komami, N.; Kojima, M.; Yoshino, T.; Matsunaga, S.
Asian J. Org. Chem. 2019, 8, 1107-1110. DOI: 10.1002/ajoc.201900200

Cobalt-Catalyzed Allylic Alkylation Enabled by Organophotoredox Catalysis
Takizawa, K.; Sekino, T.; Sato, S.; Yoshino, T.; Kojima, M.; Matsunaga, S.
Angew. Chem., Int. Ed. 2019, 58, 9199-9203. DOI: 10.1002/anie.201902509

Synthesis of Functionalized Monoaryl-λ3-iodanes through Chemo- and Site-selective ipso-Substitution Reactions
Komami, N.; Matsuoka, K.; Nakano, A.; Kojima, M.; Yoshino, T.; Matsunaga, S.
Chem. Eur. J. 2019, 25, 1217-1220. DOI: 10.1002/chem.201805970

Enantioselective C(sp3)–H Amidation of Thioamides Catalyzed by a CobaltIII/Chiral Carboxylic Acid Hybrid System
Fukagawa, S.; Kato, Y.; Tanaka, R.; Kojima, M.; Yoshino, T.; Matsunaga, S.
Angew. Chem., Int. Ed. 2019, 58, 1153-1157. DOI: 10.1002/anie.201812215
Highlighted in Synfacts.

One-Step Synthesis of 4H-3,1-Benzoxazin-4-ones from Weinreb Amides and 1,4,2-Dioxazol-5-ones via Cobalt-Catalyzed C–H Bond Activation
Tanimoto, I.; Kawai, K.; Sato, A.; Yoshino, T.; Matsunaga, S.
HeteroCycles 2019, 99, 118-125. DOI: 10.3987/COM-18-S(F)48

2018
Chiral Carboxylic Acid-Enabled Achiral Rhodium(III)-Catalyzed Enantioselective C−H Functionalization
Lin, L.; Fukagawa, S.; Sekine, D.; Tomita, E.; Yoshino, T.; Matsunaga, S.
Angew. Chem., Int. Ed. 2018, 57, 12048-12052. DOI: 10.1002/anie.201807610
Highlighted in Synfacts.

Pentamethylcyclopentadienyl Rhodium(III)-Chiral Disulfonate Hybrid Catalysis for Enantioselective C–H Bond Functionalization
Satake, S.; Kurihara, T.; Nishikawa, K.; Mochizuki, T.; Hatano, M.; Ishihara, K.; Yoshino, T.; Matsunaga, S.
Nature Catalysis 2018, 1, 585-591. DOI: 10.1038/s41929-018-0106-5
Author Correction

5-((3-Bromoallyl)Sulfonyl)-1H-Tetrazoles for Bromodiene Synthesis
Suzuki, T.; Fukagawa, S.; Yoshino, T.; Anada, M.; Matsunaga, S.
HeteroCycles 2018, 97, 1304-1312. DOI: 10.3987/COM-18-S(T)90
Weinreb Amide Directed Versatile C–H Bond Functionalization under (η5-Pentamethylcyclopentadienyl)cobalt(III) Catalysis
Kawai, K.; Bunno, Y.; Yoshino, T.; Matsunaga, S.
Chem. Eur. J. 2018, 24, 10231-10237. DOI: 10.1002/chem.201801750

Cobalt-catalyzed C(sp3)–H Bond Functionalization Reactions
Yoshino, T.; Matsunaga, S.
Asian J. Org. Chem. 2018, 7, 1193-1205. DOI: 10.1002/ajoc.201800195

Synthesis of 1,1'-Spirobiindane-7,7'-Disulfonic Acid and Disulfonimide: Application for Catalytic Asymmetric Aminalization
Kurihara, T.; Satake, S.; Hatano, M.; Ishihara K.; Yoshino T.; Matsunaga, S.
Chem. Asian. J. 2018, 13, 2378-2381. DOI: 10.1002/asia.201800341
DOCK1 inhibition suppresses cancer cell invasion and macropinocytosis induced by self-activating Rac1P29S mutation
Tomino, T.; Tajiri, H.; Tatsuguchi, T.; Shirai, T.; Oisaki, K.; Matsunaga, S.; Sanematsu, F.; Sakata, D.; Yoshizumi, T.; Maehara, Y.; Kanai, M.; Cote, J.-F.; Fukui, Y.; Uruno Y.
Biochem. Biophys. Res. Commun. 2018, 497, 298-304. DOI: 10.1016/j.bbrc.2018.02.073
Palladium-Catalyzed Germylation of Aryl Bromides and Aryl Triflates Using Hexamethyldigermane
Komami, N.; Matsuoka, K.; Yoshino, T.; Matsunaga S.
Synthesis 2018, 50, 2067-2075. DOI: 10.1055/s-0037-1609301
Highlighted in the news section on Thieme Chemistry homepage.

Synthesis of Fluorine-Containing 6-Arylpurine Derivatives via Cp*Co(III)-Catalyzed C–H Bond Activation
Murakami, N.; Yoshida, M.; Yoshino, T.; Matsunaga S.
Chem. Pharm. Bull. 2018, 66, 51-54. DOI: 10.1248/cpb.c17-00797

2017
Structural and thermodynamic analyses reveal critical features of glycopeptide recognition by the human PILRα immune cell receptor
Furukawa, A.; Kakita, K.; Yamada, T.; Ishizuka, M.; Sakamoto, J.; Hatori, N.; Maeda, N.; Ohsaka, F.; Saitoh, T.; Nomura, T.; Kuroki, K.; Nambu, H.; Arase, H.; Matsunaga, S.; Anada, M.; Ose, T.; Hashimoto, S.; Maenaka, K.
J. Biol. Chem. 2017, 292, 21128-21136. DOI: 10.1074/jbc.M117.799239
Total Synthesis of Brasilicardins A and C
Anada M.; Hanari T.; Kakita K.; Kurosaki Y.; Katsuse K.; Sunadoi Y.; Jinushi Y.; Takeda K.; Matsunaga S.; Hashimoto S.
Org. Lett. 2017, 19, 5581-5584. DOI: 10.1021/acs.orglett.7b02728

2-Hydroxyindoline-3-triethylammonium Bromide: A Reagent for Formal C3-Electrophilic Reactions of Indoles
Abe T.; Suzuki, T.; Anada M.; Matsunaga, S.; Yamada K.
Org. Lett. 2017, 19, 4275-4278. DOI: 10.1021/acs.orglett.7b01940

High-Valent Cobalt-Catalyzed C–H Bond Functionalization
Yoshino T.; Matsunaga S.
Advances in Organometallic Chemistry 2017, 68, 197-247. DOI: 10.1016/bs.adomc.2017.07.002
Cp*CoIII-Catalyzed C–H Alkenylation/Annulation Reactions of Indoles with Alkynes: A DFT Study
Sakata, K.; Eda M.; Kitaoka Y.; Yoshino T.; Matsunaga S.
J. Org. Chem. 2017, 82, 7379-7387. DOI: 10.1021/acs.joc.7b01047

Catalytic Enantioselective Desymmetrization of meso-Aziridines with Fluoromalonates
Fukagawa, S.; Xu, Y.; Anada, M.; Yoshino, T.; Matsunaga, S.
HeteroCycles 2017, 94 1337-1350. DOI: 10.3987/COM-17-13725.

Targeting Ras-Driven Cancer Cell Survival and Invasion through Selective Inhibition of DOCK1
Tajiri, H.; Uruno, T.; Shirai, T.; Takaya, S.; Matsunaga, S.; Setoyama, D.; Watanabe, M.; Kukimoto-Niino, M.; Oisaki, K.; Ushijima, M.; Sanematsu, F.; Honma, T.; Terada,T.; Oki,E.; Shirasawa, S.; Maehara, Y.; Kang, D.; Côté, J.-F.; Yokoyama, S.; Kanai, M.; Fukui, Y.
Cell Reports 2017, 19, 969-980. DOI: 10.1016/j.celrep.2017.04.016

Structural and thermodynamic analyses reveal critical features of glycopeptide recognition by the human PILRα immune cell receptor
Furukawa, A.; Kakita, K.; Yamada, T.; Ishizuka, M.; Sakamoto, J.; Hatori, N.; Maeda, N.; Ohsaka, F.; Saitoh, T.; Nomura, T.; Kuroki, K.; Nambu, H.; Arase, H.; Matsunaga, S.; Anada, M.; Ose, T.; Hashimoto, S.; Maenaka, K.
J. Biol. Chem. 2017, 292, 21128-21136. DOI: 10.1074/jbc.M117.799239
Stereoselective Synthesis of Tetrasubstituted Alkenes via a Cp*CoIII-Catalyzed C−H Alkenylation/Directing Group Migration Sequence
Ikemoto. H.; Tanaka, R.; Sakata, K.; Kanai, M.; Yoshino, T.; Matsunaga, S.
Angew. Chem., Int. Ed. 2017, 56, 7156-7160. DOI: 10.1002/anie.201703193

Cp*CoIII-catalyzed Directed C–H Trifluoromethylthiolation of 2-Phenylpyridines and 6-Arylpurines
Yoshida, M.; Kawai, K.; Tanaka, R.; Yoshino, T.; Matsunaga, S.
Chem. Commun. 2017, 53, 5974-5977. DOI: 10.1039/C7CC03072J

A Novel Spiro-heterocyclic Compound Identified by the Silkworm Infection Model Inhibits Transcription in Staphylococcus aureus
Paudel, A.; Hamamoto, H.; Panthee, S.; Kaneko, K.; Matsunaga, S.; Kanai, M.; Suzuki, Y.; Sekimizu, K.
Front. Microbiol. 2017, 7, 712. DOI: 10.3389/fmicb.2017.00712
Hybrid Catalysis Enabling Room-Temperature Hydrogen Gas Release from N-Heterocycles and Tetrahydronaphthalenes
Kato, S.; Saga, Y.; Kojima, M.; Fuse,H.; Matsunaga, S.; Fukatsu, A.; Kondo, M.; Masaoka, S.; Kanai, M.
J. Am. Chem. Soc. 2017, 139, 2204-2207. DOI: 10.1021/jacs.7b00253

(Pentamethylcyclopentadienyl)cobalt(III)-Catalyzed C–H Bond Functionalization: From Discovery to Unique Reactivity and Selectivity
Yoshino, T.; Matsunaga, S.
Adv. Synth. Catal. 2017, 359, 1245-1262. DOI: 10.1002/adsc.201700042
Selected for a cover picture, DOI: 10.1002/adsc.201700284

Diastereo- and Enantioselective Construction of 6,7-Dioxabicyclo[2.2.1]heptane Derivatives by a Dirhodium(II)-Catalyzed Intramolecular C–H Insertion Reaction
Miyazawa, T.; Imai, K.; Ito, M.; Takeda, K.; Anada, M.; Matsunaga, S.; Hashimoto, S.
HeteroCycles 2017, 95, 1211-1229. DOI: 10.3987/COM-16-S(S)72

2016
Rapid Screening by Cell-Based Fusion Assay for Identifying Novel Antivirals of Glycoprotein B-Mediated Herpes Simplex Virus Type 1 Infection
Maeda, N.; Furukawa, A; Kakita, K.; Anada, M.; Hashimoto, S.; Matsunaga, S.; Kuroki, K.; Ose, T.; Kato, A.; Arii, J.; Kawaguchi, Y.; Arase, H.; Maenaka, K.
Biol. Pharm. Bull. 2016, 39, 1897-1902. DOI: 10.1248/bpb.b16-00533

Site- and Regioselective Monoalkenylation of Pyrroles with Alkynes via Cp*CoIII Catalysis
Tanaka, R.; Ikemoto, H.; Kanai, M.; Yoshino, T.; Matsunaga, S.
Org. Lett. 2016, 18, 5732-5735. DOI: 10.1021/acs.orglett.6b02997

Enantio-and diastereoselective desymmetrization of α-alkyl-α-diazoesters by dirhodium(II)-catalyzed intramolecular C–H insertion
Miyazawa, T.; Minami, K.; Ito, M;, Anada, M.; Matsunaga, S.; Hashimoto, S.
Tetrahedron 2016, 72, 3939-3947. DOI: 10.1016/j.tet.2016.05.015

Cp*CoIII-Catalyzed Dehydrative C–H Allylation of 6-Arylpurines and Aromatic Amides Using Allyl Alcohols in Fluorinated Alcohols
Bunno, Y.; Murakami, N.; Suzuki, Y.; Kanai, M.; Yoshino, T.; Matsunaga, S.
Org. Lett. 2016, 18, 2216-2219. DOI: 10.1021/acs.orglett.6b00846

Carbon Dioxide Utilization via Carbonate-promoted C–H Carboxylation
Banerjee, A; Dick, G. R.; Yoshino, T.; Kanan, M. W.
Nature 2016, 531, 215-219. DOI: 10.1038/nature17185

2015
Catalytic Asymmetric Iterative/Domino Aldehyde Cross-Aldol Reactions for the Rapid and Flexible Synthesis of 1,3-Polyols
Lin. L.; Yamamoto, K.; Mitsunuma, H.; Kanzaki, Y.; Matsunaga, S.; Kanai, M.
J. Am. Chem. Soc. 2015, 137, 15418-15421. DOI: 10.1021/jacs.5b11192

Cp*CoIII Catalyzed Site-Selective C–H Activation of Unsymmetrical O-Acyl Oximes: Synthesis of Multisubstituted Isoquinolines from Terminal and Internal Alkynes
Sun, B.; Yoshino, T.; Kanai, M.; Matsunaga, S.
Angew. Chem., Int. Ed. 2015, 54, 12968-12972. DOI: 10.1002/anie.201507744

Dehydrative Direct C–H Allylation with Allylic Alcohols under [Cp*CoIII] Catalysis
Suzuki, Y.; Sun, B.; Sakata, K.; Yoshino, T.; Matsunaga, S.; Kanai, M.
Angew. Chem., Int. Ed. 2015, 54, 9944-9947. DOI: 10.1002/anie.201503704

Cp*Co(III)-catalyzed Oxidative C–H Alkenylation of Benzamides with Ethyl Acrylate
Suzuki, Y.; Sun, B.; Yoshino, T.; Matsunaga, S.; Kanai, M.
Tetrahedron 2015, 71,4552-4556. DOI: 10.1016/j.tet.2015.02.032

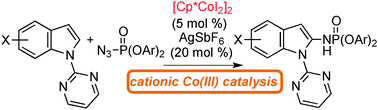
A Cp*CoI2-dimer as A Precursor for Cationic Co(III)-catalysis: Application to C–H Phosphoramidation of Indoles
Sun, B.; Yoshino, T.; Matsunaga, S.; Kanai, M.
Chem. Commun. 2015, 51, 4659-4661. DOI: 10.1039/C4CC10284C
2014
Regiodivergent Kinetic Resolution of Terminal and Internal rac-Aziridines with Malonates under Dinuclear Schiff Base Catalysis
Xu, Y.; Kaneko, K.; Kanai, M.; Shibasaki, M.; Matsunaga, S.
J. Am. Chem. Soc. 2014, 136, 9190-9194. DOI: 10.1021/ja5039165

Pyrroloindolone Synthesis via a Cp*CoIII-Catalyzed Redox-Neutral Directed C–H Alkenylation/Annulation Sequence
Ikemoto, H.; Yoshino, T.; Sakata, K.; Matsunaga, S.; Kanai, M.
J. Am. Chem. Soc. 2014, 136, 5424-5431. DOI: 10.1021/ja5008432
*Featured by SJN news.

Air-Stable Carbonyl(pentamethylcyclopentadienyl)cobalt Diiodide Complex as a Precursor for Cationic (Pentamethylcyclopentadienyl)cobalt(III) Catalysis: Application for Directed C-2 Selective C–H Amidation of Indoles
Sun, B.; Yoshino, T.; Matsunaga, S.; Kanai, M.
Adv. Synth. Catal. 2014, 356, 1491-1495. DOI: 10.1002/adsc.201301110
*Selected as cover picture.

Cobalt-Catalyzed C-4 Selective Alkylation of Quinolines
Yamamoto, S.; Saga, Y.; Andou, T.; Matsunaga, S.; Kanai, M.
Adv. Synth. Catal. 2014, 356, 401-405. DOI: 10.1002/adsc.201300991

Enantioselective Synthesis of Spirooxindoles via Direct Catalytic Asymmetric Aldol-Type Reaction of Isothiocyanato Oxindoles
Kato, S.; Kanai, M.; Matsunaga, S.
Heterocycles 2014, 88, 475-491. DOI: 10.3987/COM-13-S(S)60

2013
Rh-Catalyzed Aldehyde-Aldehyde Cross-Aldol Reaction under Base-Free Conditions: In-Situ Aldehyde-Derived Enolate Formation through Orthogonal Activation
Lin, L.; Yamamoto, K.; Matsunaga, S.; Kanai, M.
Chem. Asian J. 2013, 8, 2974-2983. DOI: 10.1002/asia.201300928
Structure-activity relationship study of novel iminothiadiazolo-pyrimidinone antimicrobial agents
Paudel, A.; Kaneko, K.; Watanabe, A.; Matsunaga, S.; Kanai, M.; Hamamoto, H.; Sekimizu, K.
J. Antibiot. 2013, 66, 663-667. DOI: 10.1038/ja.2013.69
Cp*CoIII-Catalyzed C2-Selective Addition of Indoles to Imines
Yoshino, T.; Ikemoto, H.; Matsunaga, S.; Kanai, M.
Chem. Eur. J. 2013, 19, 9142-9146. DOI: 10.1002/chem.201301505

Sultam Synthesis via Cu-Catalyzed Intermolecular Carboamination of Alkenes with N-Fluorobenzenesulfonimide
Kaneko, K.; Yoshino, T.; Matsunaga, S.; Kanai, M.
Org. Lett. 2013, 15, 2502-2505. DOI: 10.1021/ol4009848

Catalytic Asymmetric Synthesis of Spirooxindoles via Addition of Isothiocyanato Oxindoles to Aldehydes Under Dinuclear Nickel Schiff Base Catalysis
Kato, S.; Kanai, M.; Matsunaga, S.
Chem. Asian J. 2013, 8, 1768-1771. DOI: 10.1002/asia.201300251

Cobalt-Catalyzed C4-Selective Direct Alkylation of Pyridines
Andou, T.; Saga, Y.; Komai, H.; Matsunaga, S. Kanai, M.
Angew. Chem., Int. Ed. 2013, 52, 3213-3216. DOI: 10.1002/anie.201208666
Featured by SYMFORM 2013/06
Featured by ワイリー・サイエンスカフェ

A Cationic High-Valent Cp*CoIII Complex for the Catalytic Generation of Nucleophilic Organometallic Species: Directed C–H Bond Activation
Yoshino, T. Ikemoto, H.; Matsunaga, S.; Kanai, M.
Angew. Chem., Int. Ed. 2013, 52, 2207-2211. DOI: 10.1002/anie.201209226

Multimetallic Schiff Base Complexes as Cooperative Asymmetric Catalysts
Matsunaga, S.; Shibasaki, M.
Synthesis 2013, 45, 421-437. DOI: 10.1055/s-0032-1316846
2012
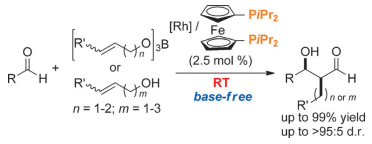
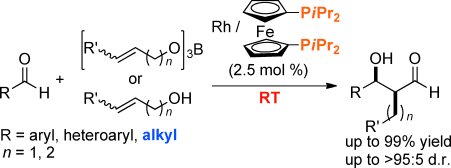
Rhodium-Catalyzed Cross-Aldol Reaction: In Situ Aldehyde-Enolate Formation from Allyloxyboranes and Primary Allylic Alcohols
Lin, L.; Yamamoto, K.; Matsunaga, S.; Kanai, M.
Angew. Chem., Int. Ed. 2012, 51, 10275-10279. DOI: 10.1002/anie.201205680
Direct Catalytic Asymmetric Vinylogous Michael Reaction of α,β-Unsaturated γ-Butyrolactam under Dinuclear Nickel Schiff Base Catalysis
Tanabe, H.; Xu, Y.; Sun, B.; Matsunaga, S.; Shibasaki, M.
Heterocycles 2012, 86, 611-622. DOI: 10.3987/COM-12-S(N)58

Enantioselective Synthesis of 2,2-Disubstituted Terminal Epoxides via Catalytic Asymmetric Corey-Chaykovsky Epoxidation of Ketones
Sone, T.; Yamaguchi, A.; Matsunaga, S.; Shibasaki, M.
Molecules 2012, 17, 1617-1634. DOI: 10.3390/molecules17021617 (open access)
Catalytic Asymmetric Amination of Oxindoles under Dinuclear Nickel Schiff Base Catalysis
Mouri, S.; Chen, Z.; Matsunaga, S.; Shibasaki, M.
Heterocycles 2012, 84, 879-892. DOI: 10.3987/COM-11-S(P)66

Catalytic Asymmetric Synthesis of Spirooxindoles by a Mannich-Type Reaction of Isothiocyanato Oxindoles
Kato, S.; Yoshino, T.; Shibasaki, M.; Kanai, M.; Matsunaga, S.
Angew. Chem., Int. Ed. 2012, 51, 7007-7010. DOI: 10.1002/anie.201203005

Mg-catalyzed Enantioselective Benzylic C–H Bond Functionalization of Isoindolinones: Addition to Imines
Suzuki, Y.; Kanai, M.; Matsunaga, S.
Chem. Eur. J. 2012, 18, 7654-7657. DOI: 10.1002/chem.201200821

Lewis Acid Catalyzed Benzylic C–H Bond Functionalization of Azaarenes; Addition to Imines and Enones
Komai, H.; Yoshino, T.; Matsunaga, S.; Kanai, M.
Synthesis 2012, 44, 2185-2194. DOI: 10.1055/s-0031-1291041

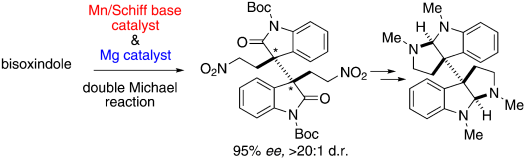
Catalytic Asymmetric Total Synthesis of Chimonanthine, Folicanthine, and Calycanthine through Double Michael Reaction of Bisoxindole
Mitsunuma, H.; Shibasaki, M.; Kanai, M.; Matsunaga, S.
Angew. Chem., Int. Ed. 2012, 51, 5217-5221. DOI: 10.1002/anie.201201132